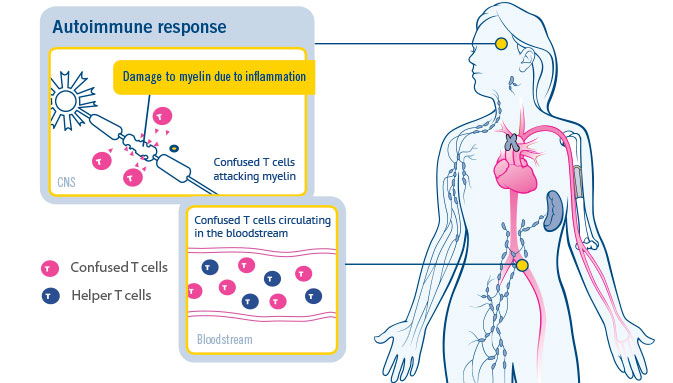
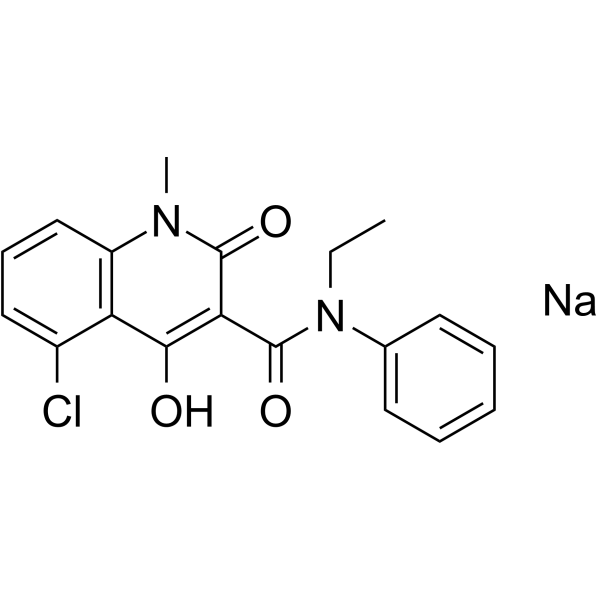
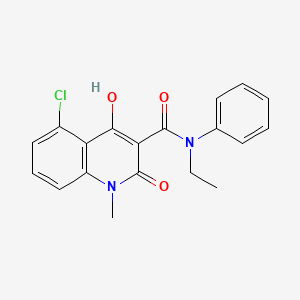
CONCERTO: A randomized, placebo-controlled trial of oral laquinimod in relapsing-remitting multiple sclerosis - Giancarlo Comi, Yuval Dadon, Nissim Sasson, Joshua R Steinerman, Volker Knappertz, Timothy L Vollmer, Alexey Boyko, Patrick Vermersch, Tjalf

Laquinimod arrests experimental autoimmune encephalomyelitis by activating the aryl hydrocarbon receptor | PNAS

The effect of laquinimod, a novel immuno-modulator in development to treat Huntington disease, on the pharmacokinetics of ethinylestradiol and levonorgestrel in healthy young women | Request PDF

Laquinimod dampens hyperactive cytokine production in Huntington's disease patient myeloid cells - Dobson - 2016 - Journal of Neurochemistry - Wiley Online Library
Laquinimod, a Quinoline-3-Carboxamide, Induces Type II Myeloid Cells That Modulate Central Nervous System Autoimmunity | PLOS ONE